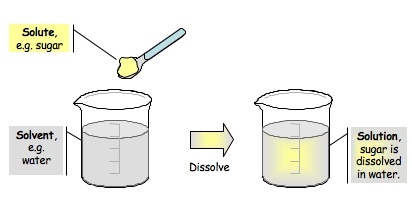
Dissolving
Forming a solution by mixing two or more materials is called dissolving. This happens because the particles are attracted to each other an always moving.
SoluteIs the substance that is being dissolved
|
SolventIs the substance that dissolves the solute
|
SolubilityThe ability for a solute to disolve in a solvent
|
SaturatedA solution that cannot hold anymore solute
|
UnsaturatedAn unsaturated solution is one in which more of the solute could dissolve at the same temperature
|
SupersaturedA supersaturated solution is when a solution which contains more solute than would normally dissolve at a certain temperature
|
The universal solvent
Water is the universal solvent because it can dissolve so many substances.
the particle model of matter and dissolving
The particle theory states that there are spaces between all particles. So, in a glass of water, there are many water particles and many empty spaces. As sugar particles seperate, the smaller water particles fit into the spaces between the larger sugar particles. The two types of particles are attracted to each other so they move close together when they are mixed.
Rate of dissolving
Rate of dissolving is how fast something dissolves. Factors that affect the rate of dissolving include agitation (or stirring), temperature (turning the heat will increase speed) and pressure.